Abstract
Background: A regimen of escalating doses of thalidomide, in combination with bortezomib and high dose melphalan (Mel/Vel/Thal) was evaluated as a conditioning regimen for autologous stem cell transplantation (ASCT) in patients (pts) with multiple myeloma (MM) in a phase I/II study.
Methods: Patients received Mel/Vel/Thal as a second of tandem ASCT if they achieved <CR to their first ASCT (tandem), or as conditioning for a salvage ASCT (salvage). Exclusion criteria were dose-intense therapy within 56 days, uncontrolled infections, severe organ dysfunction, Karnofsky score <70%, or painful grade 2 or greater peripheral neuropathy. Conditioning consisted of Vel 1.6 mg/m2 intravenously on days -4 and -1 with Mel 200 mg/m2 on day -2. Thal was given on days -5 through -1 and was administered in a planned step-wise dose escalation of 600, 800 and 1000 mg (in cohorts of 3 pts). Dexamethasone (Dex) 10-20 mg was given prior to Vel and Mel. All pts received G-CSF every other day starting day +3 until engraftment. Serious adverse events (SAEs) were graded according to CTCAE version 3.
Results: Twenty-nine pts were enrolled: 9 in the phase 1 dose-escalation phase and an additional 20 pts in phase 2. In the phase I portion, all pts experienced somnolence, with grade 3 occurring in 1 pt at the 800 mg/day dose. Subsequently, Dex 40 mg was given with first dose of Thal at the 1000 mg level with decreased severity of somnolence. No dose limiting toxicities defined as ≥ grade 4 non-hematological SAEs occurred in the phase I portion, allowing full dose escalation with 9 pts enrolled. The maximum tolerated dose for Thal was not reached and the 1000 mg dose was chosen for the phase 2 dose expansion.
No regimen related mortality occurred in either phase I or phase II portion of the study. All SAEs except lethargy and dizziness occurred after ASCT and were not attributed to Thal. The most common grade 1 and 2 non-hematologic toxicities included nausea (65.5%), mucositis (51.7%), diarrhea (48.3%), somnolence (48.3%), lethargy (27.6%), and vomiting (17.2%). The most common grade 3 non-hematologic adverse events (AEs) were neutropenic fever (58.6%), mucositis (6.9%), and somnolence (13.8%), which increased risk of falls. SAEs included somnolence (13.8%), tumor lysis syndrome (3.4%), and engraftment syndrome (3.4%). All transplant-related SAEs resolved by day +28 after ASCT. All pts achieved prompt hematological recovery with the median time to ANC >500/uL 10 days (range, 8-14 days), and platelet >20,000 12 days (range 9-26 days).
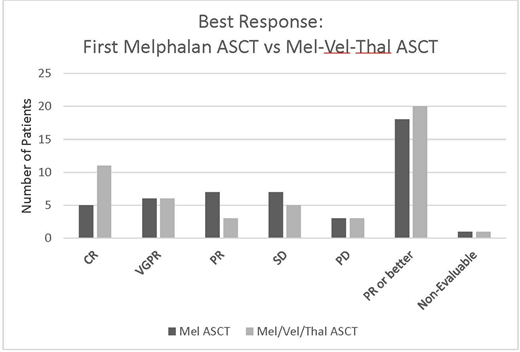
All pts received at least one ASCT prior to enrolling on the study. Seventeen pts (59%) had interim salvage chemotherapy between their upfront and Mel/Vel/Thal ASCT (i.e. received a salvage ASCT), with median time from first to salvage ASCT 29 months. The remaining 12 (41%) went directly from an upfront ASCT Mel-based ASCT to the Mel/Vel/Thal ASCT (tandem ASCT) within 6 months of the first ASCT. Twenty-seven (93%) were Durie-Salmon stage III, and 13 (44%) had >2 prior lines of therapy. Of those who had Mel/Vel/Thal as a salvage ASCT, 70% had ≥3 prior lines of therapy. The overall response rate (ORR) was 69% with 38% complete remission. ORR for Mel/Vel/Thal compared to upfront Mel ASCT was 69% versus 62% with 11 patients achieving CR with Mel/Vel/Thal compared to 5 patients with Mel alone (Figure 1). Ten of 27 evaluable patients (37%) had an upgrade in response in the Mel/Vel/Thal salvage ASCT compared to their upfront ASCT: 2 pts (7%) went from PD to PR, 1 (4%) from SD to CR, 1 (4%) from PR to VGPR; 3 (11%) from PR to CR and 2 (7%) from VGPR to CR.
Median PFS and OS were 9.3 and 65.4 months, respectively, with a median follow-up of 17.8 months. Of those who underwent tandem Mel followed by Mel/Vel/Thal ASCT the median PFS was 14.9 months with a median OS not yet reached at time of analysis. For the 17 patients who received a salvage Mel/Vel/Thal ASCT, median PFS from their upfront ASCT was 11.9 months, compared to 9.1 months with the salvage Mel/Vel/Thal ASCT.
Conclusions: High-dose Thal up to 1000 mg daily for 5 days can be safely combined with Vel and dose-intense Mel as an ASCT conditioning regimen with acceptable toxicities. Confirmation of potential synergistic effects of this combination regimen will require an appropriately designed phase III study.
Biran:BMS: Research Funding; Merck: Research Funding; Takeda: Consultancy, Speakers Bureau; Amgen: Consultancy, Speakers Bureau; Celgene: Consultancy, Honoraria, Speakers Bureau. Skarbnik:Seattle Genetics: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Abbvie: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pharmacyclics: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Gilead Sciences: Honoraria, Speakers Bureau; Novartis: Honoraria, Speakers Bureau; Genentech: Honoraria, Speakers Bureau; Jazz Pharmaceuticals: Honoraria, Speakers Bureau. Siegel:Novartis: Honoraria, Speakers Bureau; Merck: Consultancy, Honoraria, Speakers Bureau; Janssen: Consultancy, Honoraria, Speakers Bureau; Amgen: Consultancy, Honoraria, Speakers Bureau; BMS: Consultancy, Honoraria, Speakers Bureau; Karyopharm: Consultancy, Honoraria; Takeda: Consultancy, Honoraria, Speakers Bureau; Celgene: Consultancy, Honoraria, Research Funding, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal